What does a modern Clinical Research Coordinator do, and how can technology help?
Clinical Research Coordinators (CRCs) play a crucial role in the success of research sites. With a wide range of responsibilities, the CRC is the “heart” of a clinical trial—the champion of the protocol, the guardian of the patients, and the taskmaster of the research team.
Although the Clinical Research Coordinator’s purpose hasn’t changed, what they do on a daily basis has. In the past few years, most CRCs have gone from managing trials with paper and binders to managing trials with an array of different software programs.
Unfortunately, adopting software hasn’t solved all of the struggles CRCs face. In a 2021 study, 67.7% of Clinical Research Coordinators said that stress negatively impacts their work performance, even though clinical trial software is available at roughly 78% of research sites.
So what causes stress for Clinical Research Coordinators, and how can technology do a better job of removing that stress?
We interviewed several former Clinical Research Coordinators to find out what a modern CRC does, what challenges they face, and how the right technology can lessen those challenges instead of increasing them. Here’s what we learned.
What a modern Clinical Research Coordinator does
When we spoke to former Clinical Research Coordinators, they described two major components of their days: running patient visits and performing administrative tasks.
Running Patient Visits
A former Clinical Research Coordinator described her day this way:
A typical day for me started between 6:00 and 7:00 am. My first hour would be spent preparing for patient visits, checking emails, and filing paperwork. My first patient would arrive between 7:00 and 7:30, and I might see up to six or seven patients a day.
In addition to actually completing the required activities of the visit, I found myself having to document those activities in the medical record (which I had to make copies of). Then I had to transcribe them onto a paper case report form (CRF), enter them into an electronic CRF, and finally, file both the paper CRFs and the copies of the medical record in the patient’s binder.
Some visits required me to bounce back and forth between different buildings or even different campuses, holding onto those CRFs for dear life while answering phone calls from patients, PIs, sponsors, and other CRCs.
As the Clinical Research Coordinator above noted, CRCs often end up completing multiple Case Report Forms for each patient, both on paper and electronically—and that’s the real problem with participant visits.
One Clinical Research Manager we interviewed said that participant visits were the best part of her job, because participants were at the center of all she did. However, she noted that all of the paperwork that went along with participant visits was difficult to complete in a single day. This often led to her working long hours, and, in turn, to burnout.
CRCs don’t want to give up meeting with participants, but they want to minimize the amount of time they must spend on repetitive paperwork afterward. That brings us to the second time-consuming component of a Clinical Research Coordinator’s day: administrative tasks.
Performing Administrative Tasks
The same Clinical Research Coordinator described her day after patients left this way:
Once all of the patients were discharged, usually eight or nine hours later, I could return to my office to work on my administrative duties. Typically this meant scheduling and tracking patient or monitor visits or performing data entry.
Other times it could be IRB submissions, budget and contract review, or maintaining the Investigator Site File and regulatory binders. Or maybe reviewing patient records to locate potential candidates, updating study logs, or ordering supplies.
One time, I even had to chase the FedEx truck down a busy street at 8 o’clock at night to make sure records were shipped on the same day as the visit was conducted, per the study protocol. Regardless of the specific duties I was performing each day, my hours were long and my days were hectic.
The need to work after-hours and perform so much paperwork leads to Clinical Research Coordinators having a high turnover rate. The clinical research industry typically sees turnover rates of 24-29%, while the average U.S. turnover rate is 15%.
This high turnover rate doesn’t just affect individual Clinical Research Coordinators—it affects the entire clinical trial industry by making trials slower and more expensive. It takes roughly three months and costs $25,000 to train a new clinical research professional. Every time a CRC leaves their position, trials slow down and become far more expensive.
Managing Communications
One of the most pressing jobs of clinical research coordinators is to manage communication between their team, investigators, and Clinical Research Associates (CRAs). Failure to communicate clearly can lead to:
- Stress
- Conflicts between site staff and CRAs
- Unmet expectations
- Non-compliance
- Risks to participant safety
- Trial delays
- Wasted time when work has to be redone
Running patient visits while balancing administrative tasks and constant internal communication leads to heavy workloads. And those heavy workloads lead to low job satisfaction and high burnout. But compliant, efficient, and well-integrated technology might be able to help.
The workload of a modern Clinical Research Coordinator
The number of registered clinical trials taking place each year nearly doubled from 2015 to 2021. But the rate of hiring clinical research professionals hasn’t kept pace.
The number of clinical trials grew by 12.2% from 2016-2019, while job postings for clinical research professionals only grew by 9.3%.
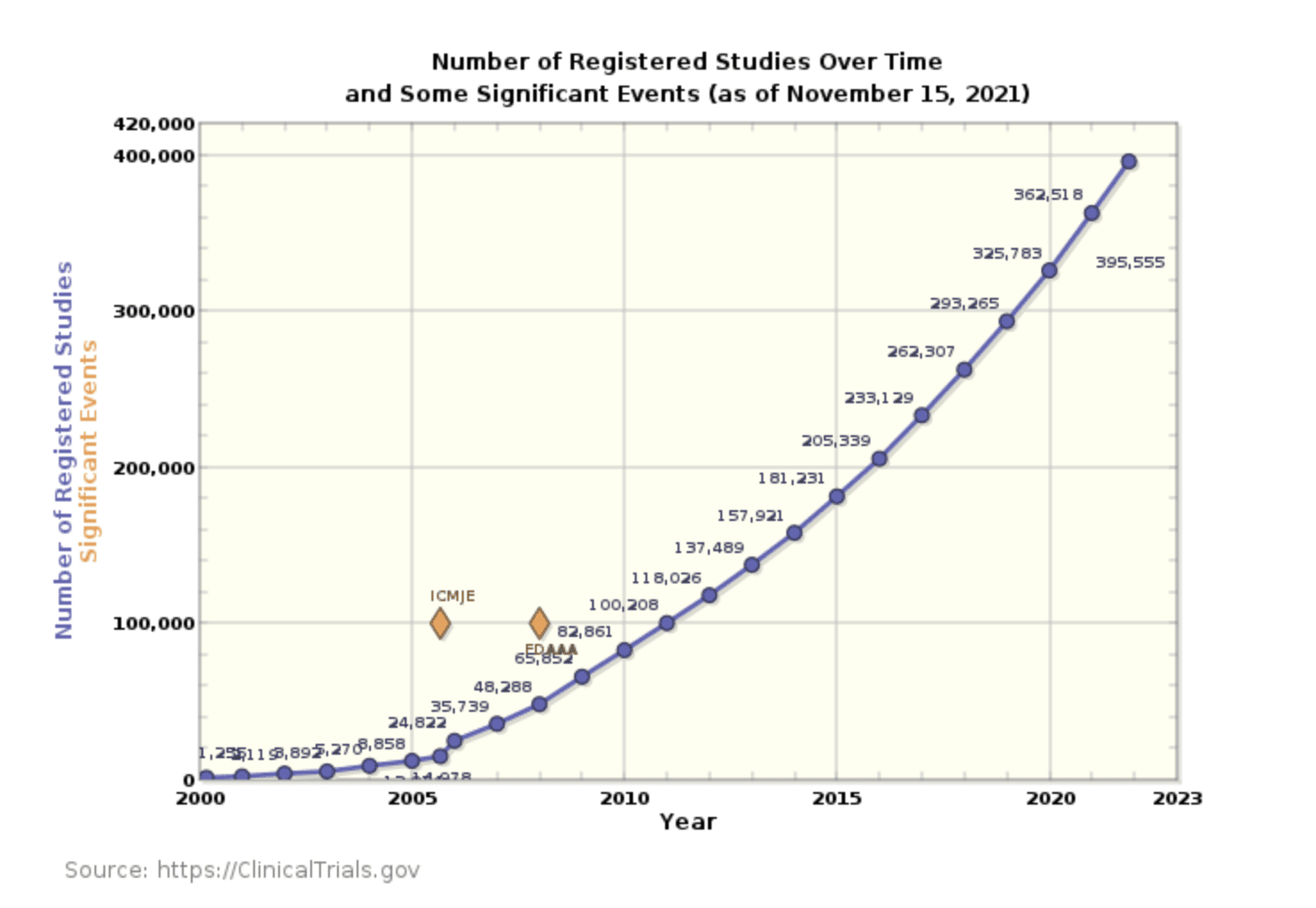
Clinical research coordinators cite this increased workload and a lack of opportunities for advancement as two major causes of job dissatisfaction. In 2020, 73.9% of Clinical Research Coordinators said they were thinking of changing jobs. They cited strict contracts, heavy workloads, and a lack of recognition as reasons why they were unhappy with their work.
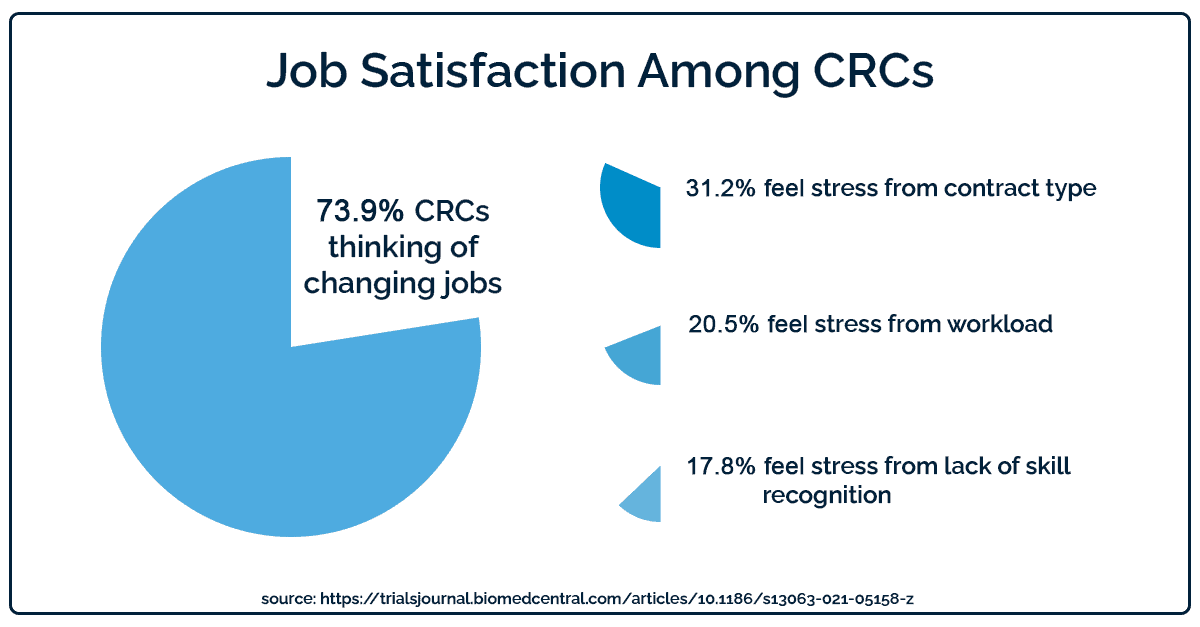
Technology can’t transform sponsor-site contracts or ensure CRCs receive recognition from their leaders. But the right software can help with heavy workloads and long hours by making CRCs more efficient.
How technology can make CRCs more efficient
A Clinical Research Coordinator’s most valuable asset is time. Streamlining research processes increases productivity, efficiency, and document quality, while also reducing CRC stress and burnout. This lets research sites hold onto their most important resource: experienced CRCs.
We’ve created this chart to show areas where CRCs could save time using technology:
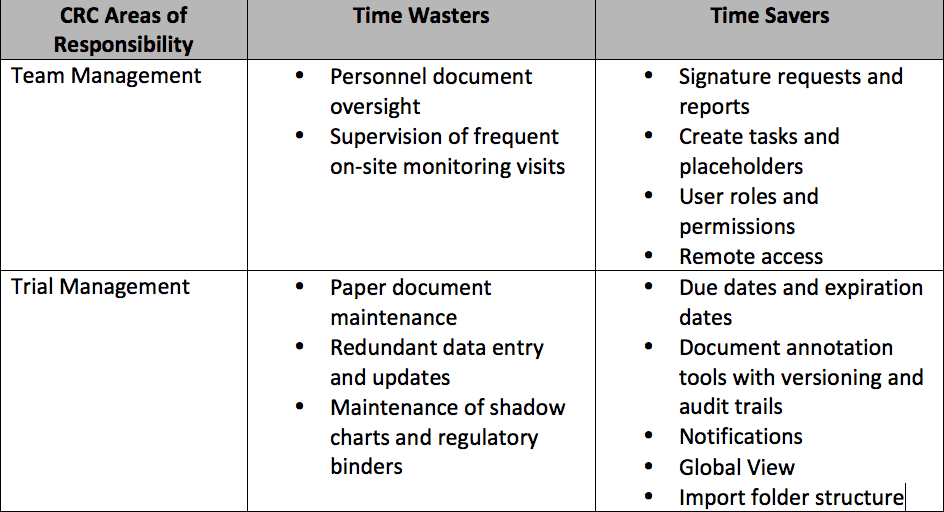
Here’s how these time-saving features work:
Team Management Features
Technology can help CRCs communicate more quickly with their team members and with monitors from sponsors or CROs.
Internally, research sites should embrace technology like a Clinical Trial Management System (CTMS) and an electronic Investigator Site File (eISF). A CTMS can automate clinical trial operations like scheduling follow-up visits for participants or managing finances.
An eISF, on the other hand, will help Clinical Research Coordinators manage regulatory documents and data collected during the study. CRCs can assign roles to members of their team to ensure everyone only sees the documents and data they have permission to see.
Internal Training and Communication
A good eISF has multiple functions. A few of those functions include letting CRCs:
- Track their own training and their team’s training for specific studies
- Create tasks and notifications for their coworkers
- Build placeholders to show their coworkers what documents are missing
All of these functions save CRCs time that would otherwise be spent on multiple emails or phone calls.
If research sites choose an eISF that allows compliant electronic signatures, CRCs can even request eSignatures from investigators and let those investigators sign from their phones or computers. No more running across a hospital campus to an investigator’s office and then waiting around for them!
With new task management software, clinical research coordinators can devote more of their time to project management and leadership and less of their time to paperwork. To learn more about using digital technology in clinical research, check out the Florence Digital Research Academy.
Trial Management Features
Trial management software helps CRCs save time during study start-up, as well as throughout the trial. A good eISF should offer trial management features like audit trails, expiration dates, and folder structures to help CRCs organize documents without relying on multiple copies.
Clinical Research Coordinators shouldn’t have to complete paper Case Report Forms and then re-enter the data into a computer. But if CRCs are going to keep only electronic copies, they need to know when those copies have been updated and by who. They also need to know when a document has expired (if it can expire) and where that document is stored.
A good eISF can help with all of these challenges. Research sites should search for eISFs with features like audit trails, expiration notifications, and clearly defined folder structures to help them keep their documents accurate and compliant.
With these features, sites can decrease how much time CRCs have to spend on document management. One of Florence’s clients sped up their document cycle time by 41.8% using an eISF with all of these capabilities.
Remote Access for Sponsors and CROs
Sharing documents and data with sponsors electronically can save Clinical Research Coordinators from chasing a FedEx truck down the street at eight p.m. But if CRCs rely on email or shared drives to share documents, they risk compliance errors.
CRCs need technology designed specifically for clinical trials. This is where remote access software comes in.
With remote access software, CRCs give their sponsor or CRO permission to view and leave comments on selected documents and data. The monitor can log into the platform to see research sites’ latest documents and source data without needing to visit the sites. This saves CRCs hours of time they used to spend mailing documents or escorting monitors around in person. It also lets monitors instantly see when site staff have updated or added documents, leading to more compliant studies, better communication, and greater trust between site staff and monitors.
Preparing for the future as a clinical research coordinator
Clinical research coordinators will always have a vital role to play in protecting patients and keeping clinical research teams organized. But if CRCs continue working overtime on repetitive tasks like re-entering data or searching for signatures, they will also continue to burn out and leave their positions at greater-than-average rates.
By retaining experienced Clinical Research Coordinators, research sites will speed up clinical trials and sponsors will save hundreds of thousands of dollars.
But to make this a reality, sites, sponsors, and CROs need to embrace technology that takes the busywork out of being a Clinical Research Coordinator. CRCs should focus on taking care of participants and using their management skills to keep trials running efficiently, not on scanning, mailing, or filing paper.
Want to learn more about becoming a Clinical Research Coordinator who focuses on serving patients and on project management instead of on paperwork? Check out our Fundamentals of Digital Research course, available for free to all clinical research professionals.

